 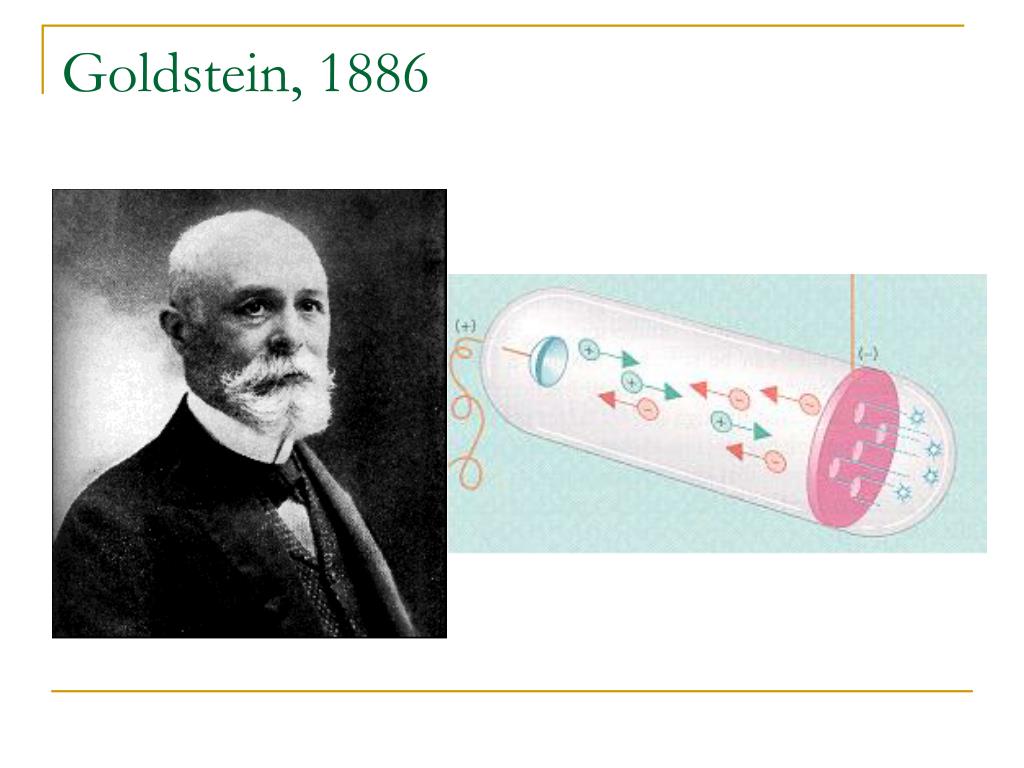
Thomson determined the charge to mass ratio of electrons.ġ898 Rutherford discovered alpha, beta, and gamma rays in radiation.ġ898 Marie Sklodowska Curie discovered radium and polonium and coined the term radioactivity after studying the decay process of uranium and thorium.ġ900 Max Planck proposed the idea of quantization to explain how a hot, glowing object emitted light.ġ900 Frederick Soddy came up with the term “isotope” to explain the unintentional breakdown of radioactive elements.ġ903 Hantaro Nagaoka proposed an atomic model called the Saturnian Model to describe the structure of an atom.ġ904 Richard Abegg found that inert gases have a “stable electron configuration.”ġ906 Hans Geiger invented a device that could detect alpha particles.ġ914 H.G.J. Goldstein discovered canal rays, which have a positive charge equal to an electron.ġ896 Henri Becquerel discovered radiation by studying the effects of x-rays on photographic film.ġ897 J.J. Stoney theorized that electricity was comprised of negative particles he called electrons.ġ879 Sir William Crookes’ experiments with cathode-ray tubes led him to confirm the work of earlier scientists by definitively demonstrating that cathode-rays have a negative charge.ġ886 E. Plucker built one of the first cathode-ray tubes.ġ869 Dmitri Mendeleev created the periodic table.ġ873 James Clerk Maxwell proposed the theory of electromagnetism and made the connection between light and electromagnetic waves.ġ874 G.J. 
Democritus’ atomic theory posited that all matter is made up small indestructible units he called atoms.ġ704 Isaac Newton theorized a mechanical universe with small, solid masses in motion.ġ803 John Dalton proposed that elements consisted of atoms that were identical and had the same mass and that compounds were atoms from different elements combined together.ġ832 Michael Faraday developed the two laws of electrochemistry.ġ859 J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
